Brazilian health regulators said Tuesday they had approved Johnson & Johnson’s experimental vaccine against the new coronavirus for the final stage of clinical trials, the fourth vaccine to receive widespread testing in the hard-hit country.
The US pharmaceutical company’s subsidiary Janssen will test the vaccine on 7,000 volunteers across seven states in Brazil, part of a group of up to 60,000 worldwide, health regulator Anvisa said in a statement.
It said the test would be a randomized, controlled, double-blind Phase 3 trial, or large-scale testing on humans — the last step before regulatory approval.
“Another vaccine study has been approved in Brazil, which is a very important development,” Anvisa official Gustavo Mendes said in a video on the regulator’s website.
Brazil has become a key testing ground in the search for a vaccine against COVID-19 since the virus is still spreading fast in the country.
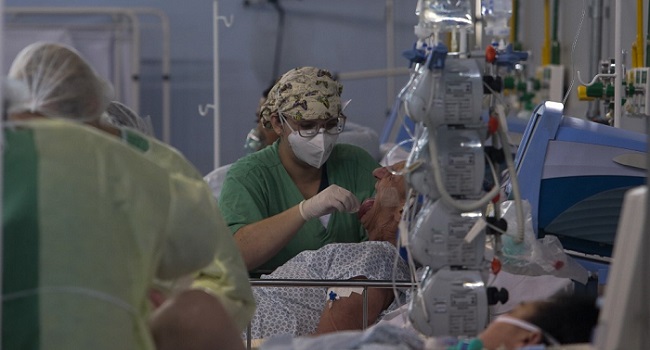
The South American nation has the second-highest number of infections and deaths in the pandemic, after the United States: nearly 3.5 million and 110,000, respectively.
Brazil has also approved three other Phase 3 trials of vaccine candidates, developed by Oxford University in partnership with pharmaceutical firm AstraZeneca, Chinese pharmaceutical firm Sinovac Biotech, and US firm Pfizer in partnership with Germany’s BioNTech.
The Brazilian state of Parana also signed a deal last week to test and produce Russia’s “Sputnik V” vaccine, which controversially became the first in the world to receive regulatory approval.
AFP